Bee Science: Varroa Mites with Dewey Caron (BSD-3)
Dr. Dewey Caron explains the biology of Varroa mites, how they reproduce inside brood cells, and why understanding their life cycle is essential for protecting honey bee colonies.
Varroa mites are the most destructive parasite affecting honey bee colonies worldwide. In this Bee Science Short, Dr. Dewey Caron explains how Varroa destructor reproduces, spreads within colonies, and why understanding the mite’s biology is essential for modern beekeeping.
Female Varroa mites enter brood cells shortly before they are capped and reproduce alongside the developing bee. Inside the sealed cell, mites feed on the developing bee and produce offspring that emerge with the young adult bee. Because the mite’s reproductive cycle is closely tied to brood production, populations can increase rapidly during the active season.
Beyond direct feeding damage, Varroa mites also spread viruses that weaken colonies and shorten the lifespan of worker bees. Dewey explains why unchecked mite populations can quickly overwhelm a colony if they are not monitored and managed.
For beekeepers, the key is awareness. Regular monitoring allows beekeepers to track mite levels and respond before populations reach damaging levels. Understanding the biology of Varroa helps beekeepers make informed decisions about management strategies and colony health.
This episode is part of the Bee Science with Dewey series, where Dr. Caron shares practical explanations of honey bee biology to help beekeepers better understand what is happening inside their colonies.
Links and references mentioned in this episode:
Lundin, Ola. Et. al. 2021. Integrated pest and pollinator management – expanding the concept. Frontiers Ecol & Environ. https://doi.org/10.1002/fee.2325
Caron, Dewey and review committee. Tools for Varroa Management. Honey Bee Health Coalition https://honeybeehealthcoalition.org/resources/varroa-management/ (you want to get the 9th edition (available soon)
Bruckner, Selina, et.al. 2025. Let’s not sugar coat it: the powdered sugar shake is Published online: https://doi.org/10.1080/00218839.2025.2550855
Caron, D.M. and L. J. Connor. 2025. Honey bee Biology and Beekeeping. Wicwas Press.
______________
Brought to you by Betterbee – your partners in better beekeeping.

Betterbee is the presenting sponsor of Beekeeping Today Podcast. Betterbee’s mission is to support every beekeeper with excellent customer service, continued education and quality equipment. From their colorful and informative catalog to their support of beekeeper educational activities, including this podcast series, Betterbee truly is Beekeepers Serving Beekeepers. See for yourself at www.betterbee.com
_______________
We hope you enjoy this podcast and welcome your questions and comments in the show notes of this episode or: questions@beekeepingtodaypodcast.com
Thank you for listening!
Podcast music: Be Strong by Young Presidents; Epilogue by Musicalman; Faraday by BeGun; Walking in Paris by Studio Le Bus; A Fresh New Start by Pete Morse; Wedding Day by Boomer; Christmas Avenue by Immersive Music; Red Jack Blues by Daniel Hart; Bolero de la Fontero by Rimsky Music; Perfect Sky by Graceful Movement; Original guitar background instrumental by Jeff Ott.
Beekeeping Today Podcast is an audio production of Growing Planet Media, LLC
** As an Amazon Associate, we may earn a commission from qualifying purchases
Copyright © 2026 by Growing Planet Media, LLC


[Bonus] Varroa Mites with Dewey (BSD-3)
Dr. Dewey Caron
Hi, I'm Dr. Dewey Caron. I come to you from Portland, Oregon. I present another audio postcard in my new series of once-monthly Beekeeping Today mini-series podcasts. Bee Science with Dr. Dewey Caron. This is the third installment in this series. Each episode seeks to blend research, field experience, and seasonal context Focusing on the why behind honeybee biology and behavior. I welcome your suggestions for timely topics.
The miniseries topic this month is mites. Yes, I know you have heard a lot about mites found in hives. Certainly Tropilaelaps and varroa mites. less perhaps about raw tracheal mites, and hardly nothing about common pollen mites. We are most interested in understanding how mites are so threatening to beekeeping success. My message is simple. If you don't get anything else from this episode, remember have a plan.
For this episode, I will be discussing varroa mites, a formidable foe to colony survival due to its involvement in spreading virus epidemics in our colonies. We are well aware that our beehives are an ideal environment for harboring the growth of pathogens with abundant, highly concentrated, stored carbohydrates, like you know, honey, and protein, bee bread. Additionally, their social organization, which fosters behavior such as food exchange and thermal regulation, among others, also aids in rapid and possible rapid possibilities passage and growth and spread. Your plan for varroa mites should be an IPM-based one. IPM is the acronym for Integrated Pest Management. Which is sometimes you might see that as IPPM for Integrated Pest and Pollinator Management The IPM mantra is one, know your enemy, two, monitor its population, three Determine the threshold level for damage the pest may cause to plant crop or host, and four, determine how best to control the pest if it exceeds an economic entry level.
IPM for varroa mite means using tools designed to kill mites in or flatten the varroa mite population growth curve. A fifth tenant is when selecting a control method, pesticides should be the last tool considered for control of the pest. But in practice, in crop and animal pest control, certainly, practitioners, practitioners being homeowners, farmers, pest control companies, beekeepers, anyone doing battle with a pest usually often reach for the bottle. The pesticide, the method that quickly and usually pretty efficient effectively will neutralize the pest, at least until pest resistance builds While we want to flatten mite population increase, we do not want to flatten the bee growth when we are using a mite control pest tool. We want the bee colony to grow and expand five to sixfold from its overwintering size. The exception is that in later spring we may need to slow colony growth if the bees start preparing to divide via swarming. That's the topic for an upcoming bee science podcast. The better we are able to flatten mite population growth in the hive, the greater our chances that we will be able to successfully meet our beekeeping objectives More honey, more efficient pollination, more potential economic return, and especially improving overwintering success.
Success can be broadly or narrowly defined as meeting both short and long-term beekeeping objectives. I'll not be going into the level of bee losses in this podcast. Thankfully, so far this winter. The extremely heavy colony losses experienced the last three to five years by US beekeepers, generally above 50% annual colony loss levels, seems to be lower Still too high, and especially for some individual beekeepers, it remains elusively high.
Heavy overwintering and annual losses tells us that we are not apparently using the best bee stock, nor Correctly mixing the number of tools that we have available to flatten mic growth expansion in our bee hives. That is where your plan comes in.
Develop a plan that will reduce mic numbers in a hive. I suggest your plan, in broad terms, might be something like improve bee health for better high survival. The devil is in the details. You'll need to have a more refined plan to succeed. So let's look at the IPM approach.
First thing I said is we need to know the enemy. So let's talk a bit about the varroa mite. The scientific name, Varroa Destructor, the species name is very apt as this might, if left unchecked, will destroy a hive. The varroa was discovered in the U. S. in 1987 after accidental introduction, probably by a beekeeper illegally bringing queens into the U. S. from Europe. The original host of varroa is an Asian honeybee. Sometime, maybe 50 to 70 years ago, it changed host to our apus mellifera. And then in the early 1970s, it was transported, again, likely by beekeepers, from Asia to Europe. It has found the European honeybee, the way we keep colonies close together in similar boxes, as both being very favorable for meeting its life's necessities.
Asian bee groom it more effectively, uncap and remove infested brood, and the mites can only develop in drone brood The Asian bee has the mite under control. Our European bee does not. Either directly or indirectly, various mites annually killed twenty five to fifty percent of beekeeper colonies, lately greater than fifty percent. When it first arrived, it killed an estimated ninety percent of non-managed feral colonies in the US.
Varroa is such a threat to European honeybees because they are a new they are a new threat European honeybees haven't had enough time to develop resistance to them. European honeybee colonies infected with varroa that are not treated will die within one to three years The varroa mite has two components to its annual life cycle. During the dispersal, formerly labeled the phoretic, Sometimes labeled the traveling phase, it switches from one adult bee host to another for an estimated average of seven days on host. It feeds primarily on the host bee's fat body, bedding itself in the first couple of intersectional membrane regions of the lower abdomen of its host adult. Nurse age bees are preferred as they have larger amounts of fat body. During spring, however, the dispersal phase is mostly spent on drones.
Because they exist in defined areas of the brood where drone brood occurs. The second phase of the mite life cycle is the reproductive phase. During this phase, a dispersal mite on a nurse bee or drone drops into a cell 15 to 18 hours prior to the cell being capped. This mite is termed a foundress or sometimes mother mite. The foundress female mite can apparently tell the bee larvae has just about finished feeding on its food provisioned in her cell. and is about to go to the next stage, what we call the prepupa. The bee prepupa spins a thin silken cocoon within her cell, while the cell is covered with a beeswax capping by adult bees. Slightly older than the nurse age bees, what we call the so-called middle-aged bees, adult bees.
The foundress mite hides in and consumes the leftover larval food at the bottom of the cell. to avoid getting trapped in the silk cocooning of the prepupa. After the prepupa completes her cocoon, it transfers into the pupil stage, and then the foundress mite climbs onto her body then opens a feeding hole in the pupal body of that worker or drone bee near the fifth abdominal segment of the bee pupa. This is needed before the bee pupa exoskeleton hardens. Only a single feeding site is open, even if multiple foundress mites invade a brood cell. The founder starts feeding on a brood within six hours of the cell being sealed. And she feeds irregularly thereafter. She feeds on fat body, which will suppress the immune system of the developing bee. This will allow for rapid development of certain viruses, which she also, in feeding, introduces into that developing bee.
The founder's mite defecates on a beeswax cell wall close to the feeding hole. This helps ID the feeding site location for her offspring. and also it becomes the place where the male mite will mate with the mature female offspring's mites. It takes sixty to seven hours after capping for that foundress mite's body to mature the first egg Which is a male egg. All remaining eggs are laid at approximately 26 to 32 hour intervals and are female eggs Five to seven eggs total may be produced within a cell by a single foundress mite. The immature mite has two immature life stages termed protonymph and deudonymph It takes about five to six days for male varroa mites to develop and seven to eight days for female mites to develop from that egg stage to a mature adult. Mating occurs in the brood self itself. The male varroa mite dies inside the shell cell shortly afterwards. And during this time it has not fed as it uses its mouth parts in mating. and so does not feed.
Reproductive output depends upon the cell entered by the foundress female. Drone brood is preferred for reproduction. And due to that two-day longer development time of the drone pupa, 14 days, compared to the 12-day capping duration of a worker cell, A drone cell yields an average of three female offspring versus a single adult female offspring. That can be produced in a worker cell. Foundress mites gradually change from drone brood of early spring to worker brood as fewer drone cells become available after the summer solstice, basically the end of June, early July. Varroa mites usually live for two months and reproduce an average of three to four times. A single foundress can produce up to 30 offspring in her lifetime. They live longer over winter, where they're between the sclerites, those hardened plates of the exoskeleton and the doubt worker bees.
Varroa mite population growth is determined by the number of female mites in the honeybee colony and the reproductive rate of female mites. As well as the availability, of course, of brood and type of brood that is available. Four very important factors that are all hard to try to put together and uh compute together A second aspect that I indicated for an IPM approach to mic control was monitoring the pest population.
The traditional means of monitoring how many mites are present in the bee colony is washing of a sample of typically 300 adult worker bees collected from the brood area. Which helps us helps ensure most bees in your sample container are nurse age bees, or counting mites falling onto a debris sticky board. Using the debris board, and many of us have purchased at one point or another or can a high bottom board that is available that uh makes use of a debris board very effectively, very efficiently, that you can remove it and do your counting, reinsert it, etc. But that means tedious counting of mites.
An alternative is to make a quick analysis of the level of mites that are falling onto the debris or sticky board. For example, saying one, there essentially I don't see any mites, two, there are some mites, or three, I see way too many mites. the three part evaluation and use that in a decision as to how to take how our present ongoing at the present time when we use the when we count to look at the sticky board, how that is doing and controlling uh our mites or using that debris, that fall of mites onto the debris board to evaluate post-treatment how a particular uh control might have been, how effective it might have been.
Destructive washing agents include alcohol or non-sudzing soap While non-destruct sampling methods to separate mites from their adult host are powdered sugar or CO2. There are a few others, but those are the four majors. Although these latter two do not immediately kill bees, that's the powdered sugar or CO2 sampling, they are still harmful adults I have a report by Sarah Bruckner and co-authors in Endnotes that describes how sugar shake or use of CO2 significantly shortens the lifespan of adult bees.
Looking and counting the number of mites in drone brood or on adult bee bodies are so highly unreliable as to be essentially worthless means of monitoring mite numbers We'd like to be able to develop drone brood sampling as a means of monitoring reproduction of raw mites in the spring, since foundress mites are highly likely to use drone brood for the reproductive mice cycle phase. But the non-random, highly clumped distribution of cells used by phylondrous mites means such sampling is unreliable. We don't have a technique to get around those negative aspects.
The gold standard is washing the bee bodies in alcohol. Any type of alcohol, such as rubbing alcohol to more concentrated alcohol, can be used. And the sample agitated for several min minutes as in use of a shaking device, for example. Repeated washings of the bee sample until no further mites are noted adds to accuracy But a precautionary note. Study by an Oregon master beekeeper for his capstone research project collected adult workers and did three hundred bee samples as three separate washes, one collection of the bees into a white container, and then took out three separate three hundred samples. He found the number of mites recovered varied widely, sometimes as much as a threefold difference Just from the same sample, but three different samples taken. So, although there are alternative ways to monitor a pest as serious as varroa mites, some are easier and more accurate than others. Your plan should include how you will monitor.
That brings up a major issue. How should we monitor for mites considering seasonal and life cycle differences of varroa? In the spring, the majority of varroa mites, up to 70% in some estimates, will occur in drone brood. Using a sample of 300 adult worker bees does not reliably sample nor adequately represent the mite population in your beehive. We get lots of zeros or one to two mites washing adult bees in March through June. Likewise, uncapping drone brood using a capping scratcher yields highly variable results.
Once drone brood rearing slows and the reproductive mites move to worker brood, for example, in June and July after the summer solstice. Reproducing mites may be only around 50% of the total mite population, so sampling of the nurse-age worker bees is a better means of estimating the mite population size. Not necessarily great, but at least better.
Now we may recover more than three mites in a 300 bee sample Three mites equals a one percent infestation level. Higher mites then give us a fair approximation of percent infestation level of the adult bee population. The acceptable level to use in making a decision has come down over the years. At one time, 10% was considered acceptable, 10% level of might. Then it was lowered to 5%.
Our tools for Varroa for Varroa Management Guide has documented this lowering acceptance over the eight editions. we have put out of the guide. The ninth edition, now being formatted and ready for publication, lists 1% Or when colonies are large in late summer, a 2% mite level. That would be with the range of 3 to no more than 6 mites recovered in alcohols in the alcohol sample are our target goals. Colonies with these minimal levels are considered at less risk of dying in the later fall or winter period.
The colonies are keeping the mites below uh a harmful level, the colonies or our control methods. This is a bit of a misnomer. It is is in reality not to number the percent of mites that is what is important. Because the mites don't kill bees. It is the viruses of, for example, the DW bee and paralysis viruses complexes that are the killers. By washing bee bodies of mites, we are not measuring virus levels. We are only looking at the percentage of host bees that have mites.
During the winter, how might we monitor mite numbers on the overwinter adult host? Open and colony collect bees can be harmful, and debris boards are not reliable as mites are not moving host to host. They remain over the winter embedded in the abdominal sclerites of hosts. So we opt to treat prophylactically, without knowing mite population levels. We even have a special tool, oxalic acid, applied as drizzle or by our vaporization, that is when we heat the crystals to the gaseous form. That can be used. Taking advantage of the lack of, or because there is a reduced breeching at this time. Our rationale is we want to get the number of mites to be as low a number as possible So the surviving foundress mite numbers are really low as they start the colony expansion in the spring. When these low numbers start reproducing, it will take longer for the mites to build up their population numbers. I. e. , we're planning to flatten the mite growth curve. Low as we can go.
Likewise, what should we use to sample early season mite numbers and the level of reproductive success? We would want to determine how rapidly they are reproducing and expanding their population. At this time, washing adult bodies is not reliable, nor is sampling drone brood. Could we look on drone adults and count how many drones have a mic? Or how many mites are present on the average drone? If say 2 or 5% of a sample of 100 adult drones have one or more mites, Could that help us determine a risk where higher numbers would key a management decision and a lower number would help reassure us that things are progressing okay? At least until next time we sample, which might be next week, next month.
We would need to look very carefully because on adult bodies the dispersal bites embed between those ventral abdominal sclerites Sometimes only a tiny sliver of their abdomen may protrude, so we may see and count them. At this point, truth in advertising, we don't have such a percentage or average number per drone that we might use as a population monitoring tool. That might be very handy if we did. Therefore, once again, we are often will apply a control without first monitoring our prophylactic approach to mic control. That's not an IPM approach.
On to the third aspect of I indicated the tenets of IPM And that is determining a threshold. IPM seeks to determine a threshold, an EIL, which stands for economic injury level of a pest infestation. The EIL threshold is the point at which the pest causes an economic difference in yield for example, for a crop, or stress that might affect livestock animal production. If the pest numbers are below this number, any negative effects of the pest infestation would not be measurable. Does not mean there would not be a negative effect, but any effect would be within a normal level that yield negative is would not be measurable. Once this threshold is reached, it will then cost time and money to negate economic injury. Cost and time to reduce the pest population below the EIL will obviously be relative, depending on what and how control is performed.
So we have a new population level, an EIT. EIT stands for economic injury threshold, would be of importance. For very minor damage above the in injury level, it might not be worth the time or cost to implement a control. If the pest population is exploding, increasing in an exponential fashion, rapid control implementation, and using a highly effective means to quickly reduce that hu the exponentially growing pest population would be called for. Crop growers employ pest control technicians to monitor pest populations. visually, using traps, computer models, or their intuition to know how rapidly to apply a control and what tools might be best to use. The tool is usually a pesticide, but remember IPM should mean other controls might be utilized and pesticides used only as a last resort.
So what are the threshold numbers beekeepers might use in their decisions on control? Now we get into murky territory. You'll read that numbers that exceed 1, 2, 3%, not so many years ago, 5, 10%, can be a used to measure risk, i. e. , they've been called a threshold. But how did we come up with these numbers? They were guesstimates from a consensus of experts.
There's virtually no science to point to why 1% or 2% or 3% are the correct threshold. And certainly times a year, 1% might be appropriate, others maybe three. They are an approximation of a threshold. Once again, recall that varroa mites are only a vector for viruses We don't need to know what that X particles of virus XYZ is cause for concern and below a level of risk. We do not need to know the number of mites, but that's what we're measuring. Well, look at the time. I would like to keep our bee science minutes below 20 minutes. Next time, let me start a discussion of spring management of the two critical populations we need managed, the honeybee and the pest varroa.
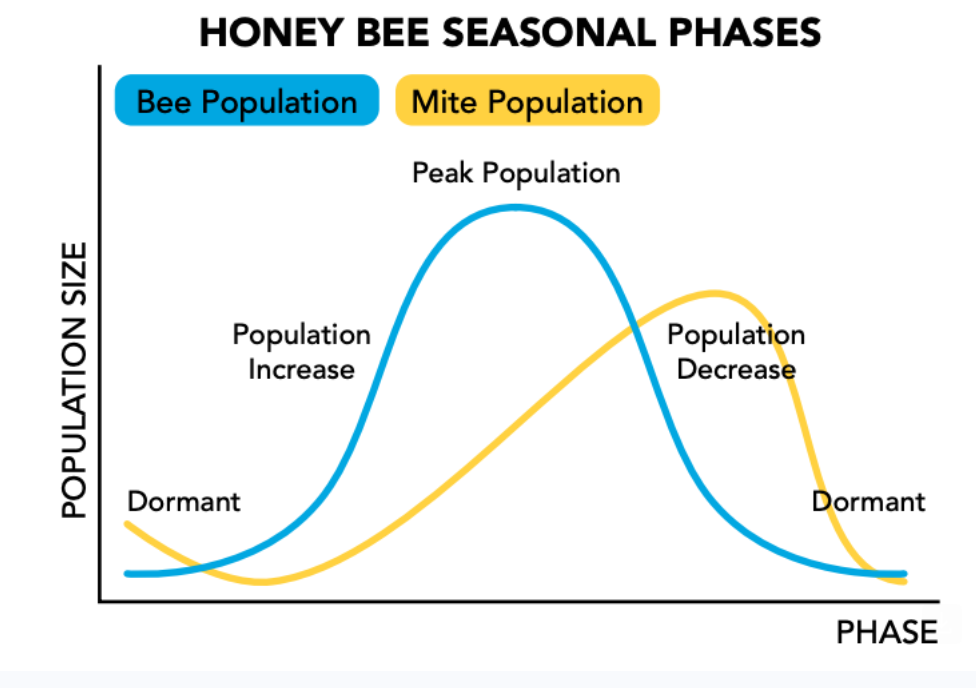
By talking about mic control, mic control will range all the way from prevent measures to use of a pesticide tool So that will be next month. With this recording, I have used a visual. The visual will show the bell-shaped curve of the bee, the honey bee population. Could be adults, it could be brood, it could be uh number drones, whatever. Estimation of the bees, number of bees. And also an estimation of the number of mites. You'll notice that the bell-shaped curve of adult worker bees, for example, is different from the skewed bell-shaped curve of mite numbers. Might numbers peak after The peak of the bee population occurs and during the period when the bee population is actually decreasing. These are the two populations we want to manage. This is what we talk about lowering or flattening that growth curve.
So this month I am asking, what is your plan? Spring is just around the corner. How are you planning to keep the low female mite numbers present in early spring colony from rapidly increasing. I'm suggesting an IPM plan. Knowing your enemy monitoring numbers and the success of their reproduction leading to that might population growth. And three, using an approximation of risk should be part of your plan. We need to keep our eye on the ball here since ultimately it is not the varroa mite itself, but their spread of virus that will constitute a success in meeting our beekeeping objectives. The last part, selecting appropriate controls, will ee up next month. Stay tuned. Be well.

PhD, Professor Emeritus, Author
Dr Dewey M. Caron is Emeritus Professor of Entomology & Wildlife Ecology, Univ of Delaware, & Affiliate Professor, Dept Horticulture, Oregon State University. He had professional appointments at Cornell (1968-70), Univ of Maryland (1970-81) and U Delaware 1981-2009, serving as entomology chair at the last 2. A sabbatical year was spent at the USDA Tucson lab 1977-78 and he had 2 Fulbright awards for projects in Panama and Bolivia with Africanized bees.
Following retirement from Univ of Delaware in 2009 he moved to Portland, OR to be closer to grandkids.
Dewey was very active with EAS serving many positions including President and Chairman of the Board and Master beekeeper program developer and advisor. Since being in the west, he has served as organizer of a WAS annual meeting and President of WAS in Salem OR in 2010, and is currently member-at-large to the WAS Board. Dewey represents WAS on Honey Bee Health Coalition.
In retirement he remains active in bee education, writing for newsletters, giving Bee Short Courses, assisting in several Master beekeeper programs and giving presentations to local, state and regional bee clubs. He is author of Honey Bee Biology & Beekeeping, major textbook used in University and bee association bee courses and has a new bee book The Complete Bee Handbook published by Rockridge Press in 2020. Each April he does Pacific Northwest bee survey of losses and management and a pollination economics survey of PNW beekeepers.
























 emerge with the young adult bee. Because the mite’s reproductive cycle is closely tied to brood production, populations can increase rapidly during the active season.
emerge with the young adult bee. Because the mite’s reproductive cycle is closely tied to brood production, populations can increase rapidly during the active season.